Steel
Based on our long-term projected MatCalc Engineering database development of ME-Fe, we can simulate precipitation evolution of a variety of steel grades, including microalloyed steels, tool steels, martensitic and maraging steels, as well as special steel grades of particular interests of our clients.
Some examples demonstrating our competence in the steel process simulation include:
Microsegregation in microalloyed steel is studied in the Scheil-Gulliver module of MatCalc. It is used to predict primary precipitates during the continuous casting process. Primary precipitates in microalloyed steel, which are marked by circles, are visible in Figure 1.

Figure1: Microstructure – primary precititates
In the calculation, back diffusion of carbon and nitrogen as well as the peritectic reaction is considered. The results can be seen in Figure 2. Primary (Ti,Nb),(C,N) precipitates with a phase fraction of 0.1% are predicted.
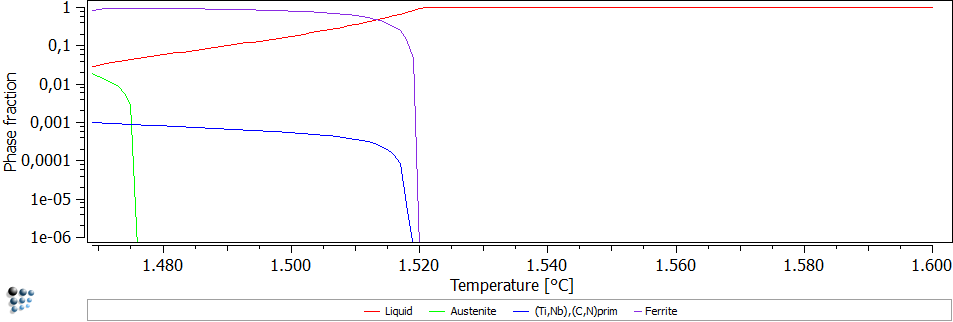
Figure2: Scheil Gulliver simulation of solidification
Transverse cracking on the surface of as-cast products is a long-standing problem in continuous casting of steel. Surface cracks occur mainly in the bending and straightening operations in a curved type continuous caster in a temperature range of 600°C to 1200°C. The reasons for this are either strain concentrations at film-like ferrite along austenite grain boundaries and/or precipitation of second phase particles. A high density of fine precipitates leads to (local) precipitation hardening with intensive stress concentrations, thus triggering the nucleation of wedge-type cavities as well as favouring interconnection of cavities surrounding the precipitates (Figure 1). This makes up the main mechanism of reduced ductility in the austenitic high-temperature region.

Figure1: Wedge-type cavities in microalloyed steel.
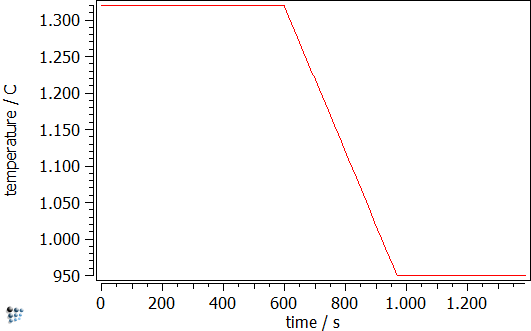
Experimentally, the probability that steel might suffer from transverse cracking in the course of the straightening operation during continuous casting is commonly assessed by hot tensile tests in a thermo-mechanical simulator (e.g. Gleeble). Thermokinetic simulations with MatCalc can predict the precipitation state, which corresponds to the particular heat treatment state. With this information, it is possible to estimate the susceptibility of transverse cracking. In the simulation, the complete thermo-mechanical history of the material, including the strain rate of deformation is considered.
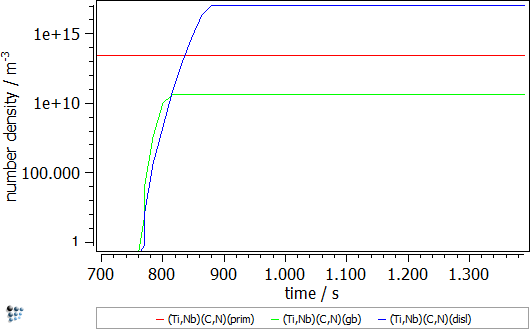
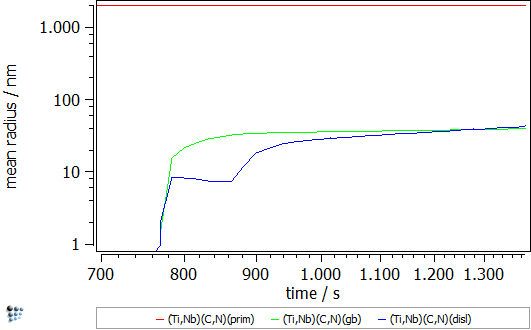
The simulation starts with the cooling segment in the continuous casting process. Afterwards follows the heat treatment, which is performed on the Gleeble. The specimens are solution treated at 1320°C and, then, cooled down to test temperature, where tensile deformation is applied. The simulated strain rate is 1∙10-3s-1 and the simulation is stopped once the specimens have been fractured in the experiment. Figure 2 displays the calculated evolution of the mean particle radii and the number densities during the Gleeble heat treatment.
Figure2: Calculated evolution of the mean particle radius and the number density of (Ti,Nb),(C,N) precipitates.
In this example, the yield strength simulation in binary Fe-Cu is shown. The strength of a material should be calibrated in a “simple” system and, then, the parameters can be transferred to complex alloys. The precipitation of Cu in Fe-Cu is based on the data of Goodman et al. (1973). It starts off with the nucleation of coherent bcc-Cu and evolves with the transformation to the semi-coherent 9R structure and the incoherent fcc-Cu. A direct-particle transformation is used for the evolution of Cu-precipitates. Figure 1 displays the calculated evolution of the mean particle radii, the number densities and the phase fractions of the different stages of Cu precipitates.
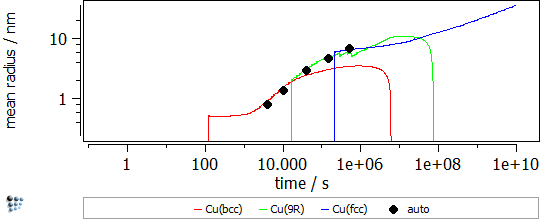
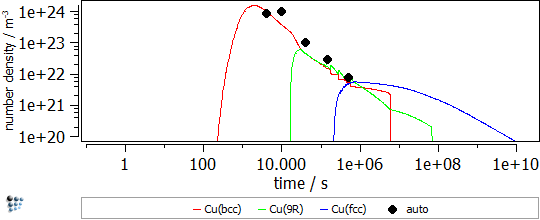
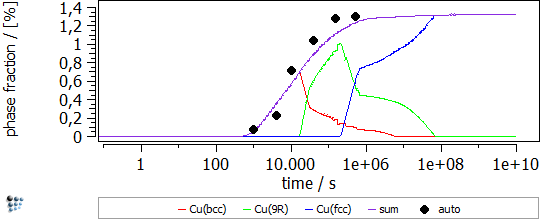
Figure1: Calculated evolution of the mean particle radii, the number densities and the phase fractions of Cu precipitates
In Figure 2 the particle strengthening contribution to the yield strength is visible (red line). The other contributions, like work hardening, intrinsic lattice strength, solid solution strengthening, grain size strengthening and subgrain size strengthening, are considered for the green line.
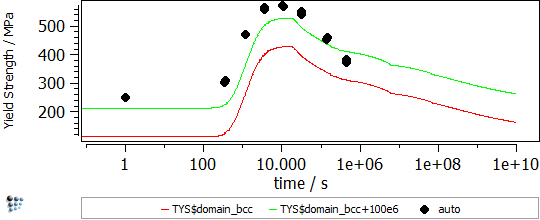
Figure2: Yield strength in binary Fe-Cu
In this example, the growth and coarsening behavior of MnS precipitates for three electrical steel grades during hot deformation is investigated. The specimens were solution treated at 1300°C for 30min and afterwards immediately cooled to test temperature and a constant stress was applied. Precipitation of MnS occurs simultaneously on grain boundaries and dislocations.
Figure 1 and 2 show the calculated radius versus time curves of MnS precipitation in three different alloys at temperatures of 800, 900 and 1000°C. Figure 1 summarizes simulation results for MnS precipitates located at grain boundaries, whereas precipitates at dislocations are shown in Figure 2.
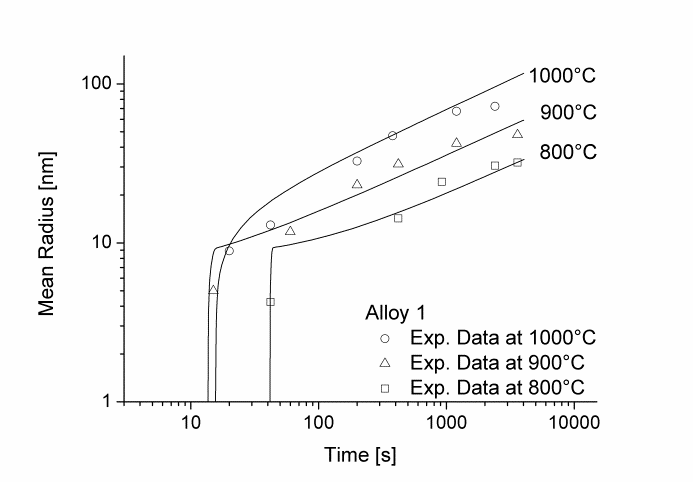
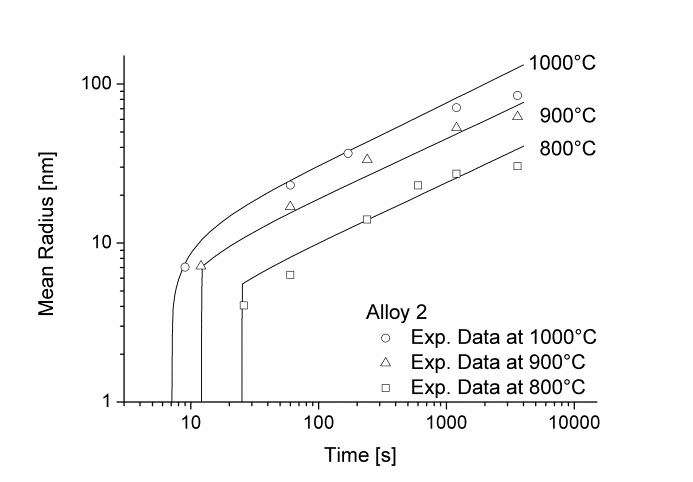
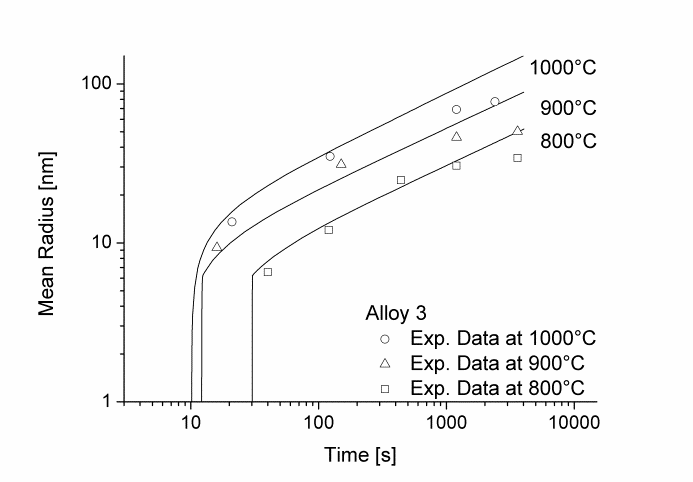
Figure1: Calculated and experimental mean radii of MnS located at grain boundaries for various temperatures
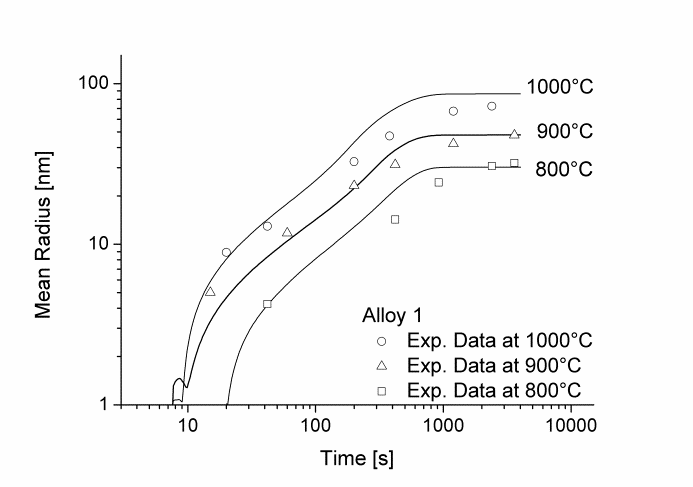
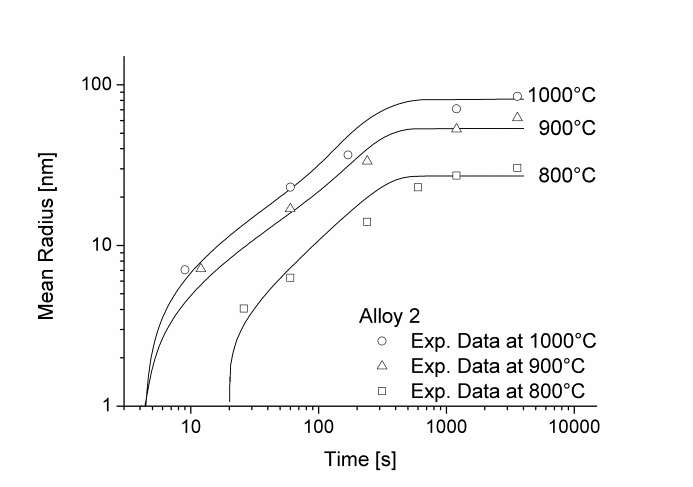
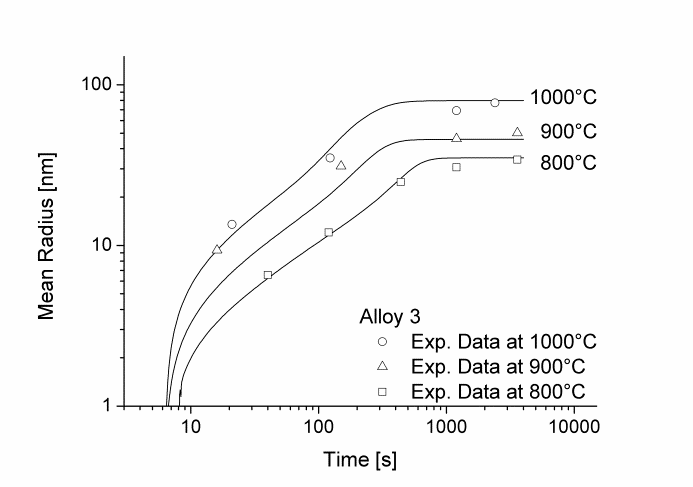
Figure2: Calculated and experimental mean radii of MnS located at dislocations for various temperatures
Figure 1 shows a Transmission Electron Microscopy (TEM) image of carbides (dark contrast) in quenched and tempered steel. The elongated carbides occur on lath-boundaries and are identified as cementite by the use of electron diffraction. The estimated size of martensite laths is related to the quenching process.

Figure1: TEM bright field image of a thin foil.
A Scanning TEM image shows contrast related to different atom masses of the same region (see Fig. 2). The chemical composition of the particle marked as “EDS spot 1” is a Cr-rich carbide. Its EDX spectrum is shown in Fig. 3.

Figure2: STEM HAADF image (mass contrast) of the same region as Fig, 2.
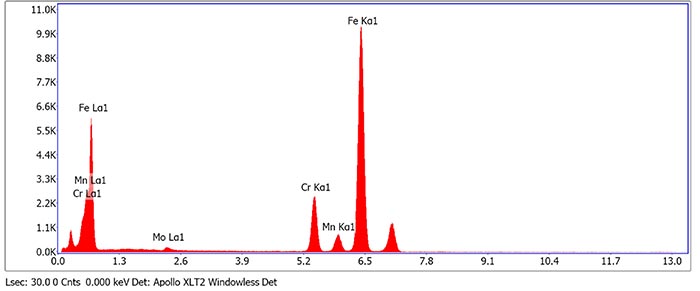
Figure3: EDX spectrum of the particle marked as "EDS Spot 1" in Fig. 2. In addition to Cr and Fe the carbide also dissolves Mn and Mo.
A complex heat treatment leads to a multimodal distribution of TiN precipitates in microalloyed steel. The figure below shows a Scanning Transmission Electron Microscopy (STEM) image with mass contrast (HAADF; the heavier, the brighter) of these particles. In this particular case the precipitates are extracted from the Fe matrix onto a thin carbon film (replica). Using this technique we are able to analyze particles down to several nanometers in size without disturbing contribution of the Fe matrix. Fig. shows three size populations of TiN precipitates. The chemical compositions of the nanoparticles were estimated using Energy Dispersive X-Ray Spectroscopy (EDX) and Electron Energy Loss Spectroscopy (EELS). The measurement of the sizes, chemical compositions and frequency of each population allows a better understanding of nitride precipitation and optimizing of the heat treatment.
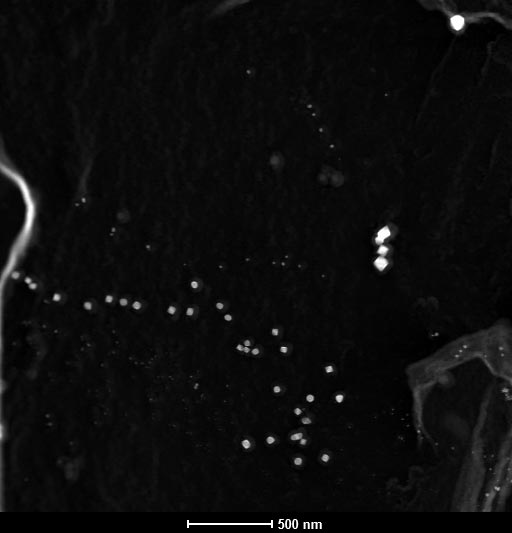
STEM HAADF image of TiN precipitates in microalloyed steel (carbon extraction replica).
We simulate the microstructure-precipitation inter-relations in alloys, using physical and semi-empiric models for dislocation substructure development, grain growth and recrystallization.
The simple example in the figure below shows the importance of excess dislocations formed during deformation for the kinetic simulation results: An increase of dislocation density of two orders of magnitude in tortion-tested microalloyed austenite relative to unstrained material is expected and leads to a perfect reproduction of the precipitation evolution during isothermal heat treatment.
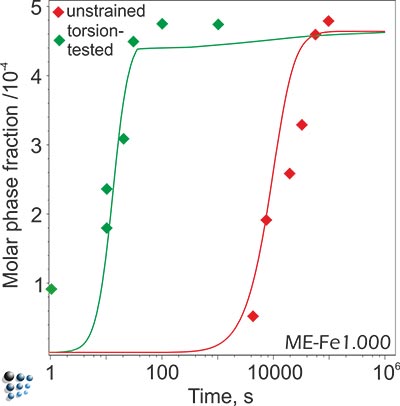
Phase fraction evolution of NbC precipitate in microallyed steels with and without deformation. The dislocations introduced during the deformation accelarate the precipitation kinetics, both by increasing the number of nucleation sites (in this system, NbC nucleates mainly on dislocations) and by diffusion enhancement.
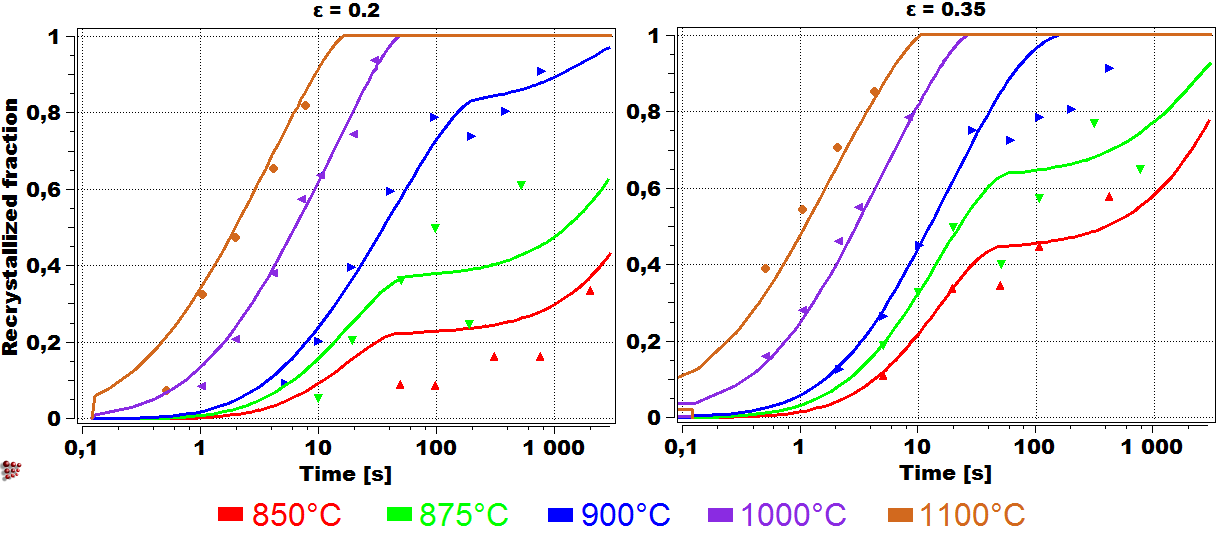
A simulation of the recrystallization progress in V-microalloyed steel for two strain values of 0.2 (left) and 0.35 (right) in compare with experimental findings (points). Recrystallization retardation by small precipitates of vanadium nitride is predicted for lower temperatures.
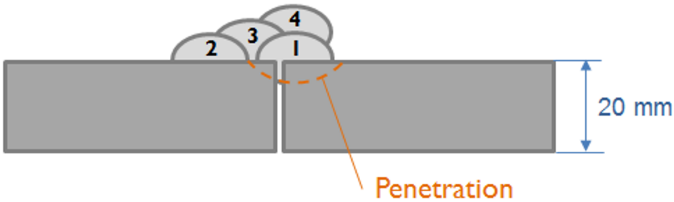
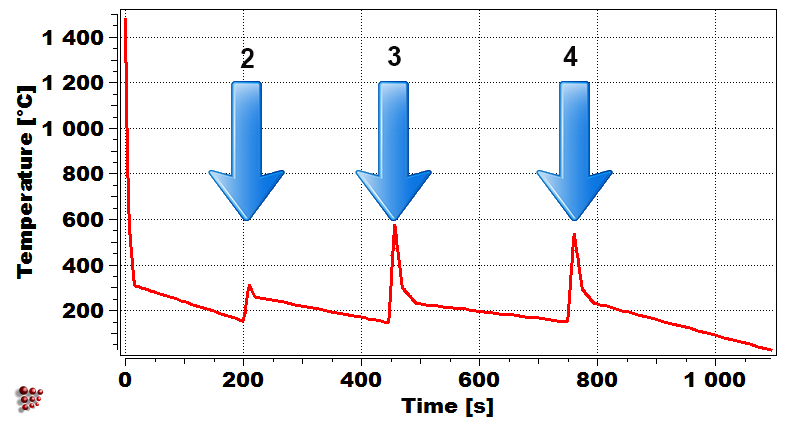
Weld bead configuration First droplet exposed temperature profile
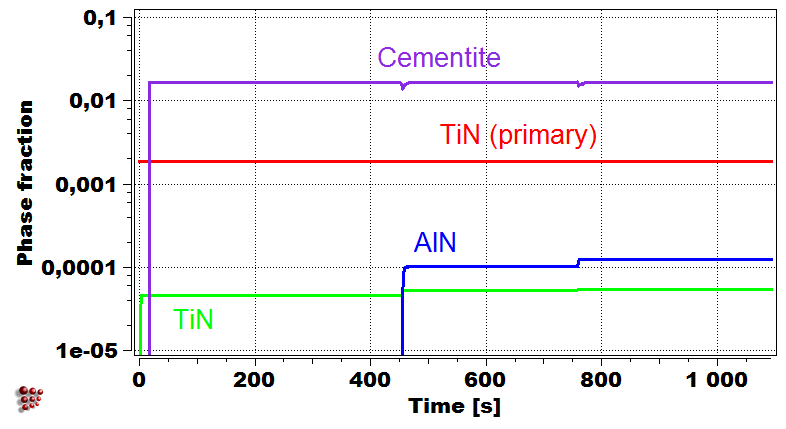
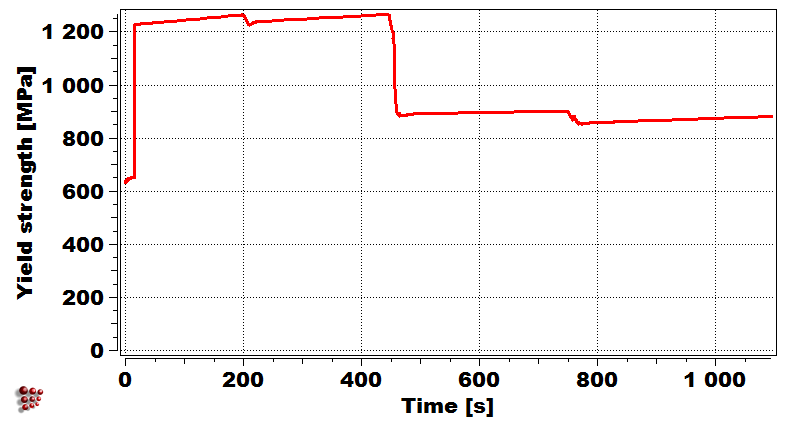
Evolution of precipitate phases in the first droplet Yield strength of first droplet during welding
Simulation of the yield strength of the first droplet of the filler material (Cr-Mo microalloyed steel) exposed to heat treatment effect of the other covering droplets.